Periodic Table GCSE: Complete Guide for Students (Easy Revision Tips)
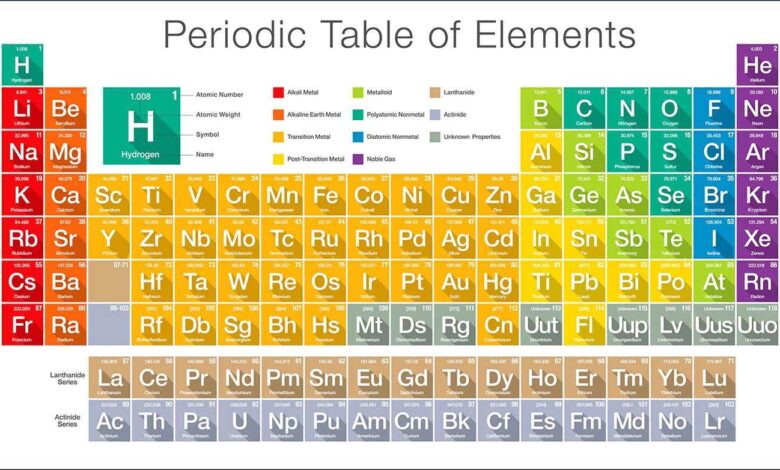
The Periodic Table GCSE is one of the most important tools in GCSE Chemistry, acting as a roadmap that organizes all known elements based on their properties and atomic structure. For students, understanding the periodic table GCSE syllabus is not just about memorizing symbols—it’s about recognizing patterns, predicting reactions, and building a strong foundation for advanced scientific concepts. Whether you’re preparing for exams or trying to grasp chemistry in everyday life, the Periodic Table GCSE plays a central role.
Historically, the periodic table was developed by Dmitri Mendeleev, who arranged elements based on their atomic mass and noticed repeating patterns in their properties. Today, the modern periodic table is arranged by atomic number, which provides a clearer and more accurate structure. This evolution highlights how scientific understanding improves over time, something that GCSE students are expected to appreciate as part of their studies.
Learning the Periodic Table GCSE is essential because it connects multiple topics in chemistry, including atomic structure, bonding, and chemical reactions. It allows students to predict how elements behave without memorizing every detail. This guide will walk you through everything you need to know, from basic structure to exam strategies, helping you approach your GCSE exams with confidence and clarity.
Structure of the Periodic Table GCSE
The Periodic Table GCSE is arranged in a highly organized way that reflects the properties and behaviors of elements. Each element is placed according to its atomic number, which represents the number of protons in its nucleus. This arrangement ensures that elements with similar characteristics appear in the same columns, making it easier to identify trends and relationships.
Rows in the Periodic Table GCSE are called periods, while columns are known as groups. Periods indicate the number of electron shells an atom has, and as you move across a period, elements gradually change from metals to non-metals. Groups, on the other hand, contain elements with the same number of electrons in their outer shell, which explains why they exhibit similar chemical properties.
Understanding this structure is key to mastering the periodic table GCSE topic. For example, elements in Group 1 are highly reactive metals, while those in Group 7 are reactive non-metals. Group 0 elements, also known as noble gases, are largely unreactive. Recognizing these patterns allows students to predict behavior without needing to memorize every element individually.
Atomic Structure and the Periodic Table GCSE
At the heart of the Periodic Table GCSE lies atomic structure, which explains why elements behave the way they do. Every atom is made up of protons, neutrons, and electrons. The number of protons determines the element, while electrons—especially those in the outer shell—play a key role in chemical reactions.
Electron configuration is particularly important in understanding the periodic table GCSE syllabus. Electrons are arranged in shells around the nucleus, and the number of occupied shells corresponds to the period of the element. Meanwhile, the number of electrons in the outer shell determines the group. This relationship makes it easier to understand why elements in the same group react similarly.
The connection between atomic structure and chemical properties is crucial. Elements tend to react in ways that allow them to achieve a stable electron configuration, often by gaining, losing, or sharing electrons. For example, Group 1 elements lose one electron to form positive ions, while Group 7 elements gain one electron to form negative ions. This predictable behavior is what makes the periodic table such a powerful tool.
Key Groups in the Periodic Table GCSE
Certain groups in the Periodic Table GCSE are especially important for GCSE students. Group 1, known as the alkali metals, includes elements like lithium, sodium, and potassium. These metals are highly reactive, especially with water, and their reactivity increases as you move down the group. This is due to the outer electron being further from the nucleus, making it easier to lose.
Group 7 elements, called the halogens, include fluorine, chlorine, and bromine. These are reactive non-metals that readily gain an electron to form negative ions. Interestingly, their reactivity decreases as you move down the group, which is the opposite of Group 1. This contrast is often tested in exams, so understanding the reasons behind these trends is essential.
Group 0 elements, or noble gases, are unique because they are very unreactive. This is due to their full outer electron shells, which makes them stable and unlikely to form bonds. These gases, such as helium and neon, are used in applications like lighting and balloons. Understanding these key groups helps students quickly identify patterns and answer exam questions more effectively.
Periodic Trends and Patterns
One of the most valuable aspects of the Periodic Table GCSE is the ability to observe trends. Across a period, atomic size decreases while reactivity and electronegativity can change significantly. Elements transition from metals on the left to non-metals on the right, showing a gradual shift in properties.
Down a group, the atomic radius increases because additional electron shells are added. This affects reactivity, as outer electrons are further from the nucleus and less strongly attracted. In metals, this means increased reactivity down the group, while in non-metals, reactivity decreases.
Understanding these trends allows students to predict properties such as boiling points, reactivity, and conductivity. Instead of memorizing isolated facts, you can rely on patterns, which is a key skill assessed in GCSE exams. This approach not only saves time but also deepens your understanding of chemistry.
Metals, Non-Metals, and Metalloids
The Periodic Table GCSE is broadly divided into metals, non-metals, and metalloids. Metals are typically found on the left side and are known for their conductivity, malleability, and shiny appearance. They tend to lose electrons during reactions, forming positive ions.
Non-metals are located on the right side of the table and have very different properties. They are generally poor conductors of heat and electricity and tend to gain electrons to form negative ions. Examples include oxygen and sulfur, which play vital roles in biological and chemical processes.
Metalloids lie between metals and non-metals and have mixed properties. They can act as semiconductors, making them important in electronics. Understanding these categories helps students quickly identify the nature of an element and predict how it will behave in a reaction.
Transition Metals (GCSE Overview)
Transition metals are found in the central block of the Periodic Table GCSE and include elements like iron, copper, and zinc. These metals have unique properties, such as high melting points, good conductivity, and the ability to form colored compounds.
One of the defining features of transition metals is their ability to form ions with different charges. This makes them versatile in chemical reactions and useful in industrial applications. For example, iron is used in construction, while copper is widely used in electrical wiring.
In GCSE Chemistry, students are expected to understand the general properties of transition metals rather than memorize specific details about each element. Recognizing their position and characteristics helps in answering questions related to bonding, reactions, and practical uses.
How to Revise the Periodic Table for GCSE Exams
Effective revision is key to mastering the periodic table GCSE topic. One of the best strategies is to use visual aids, such as diagrams and color-coded tables, to reinforce your understanding. Flashcards can also be helpful for memorizing element symbols and key properties.
Practicing past exam questions is another essential technique. This helps you become familiar with the types of questions that are likely to appear and improves your ability to apply knowledge under exam conditions. Focus on understanding trends rather than rote memorization.
Avoid common mistakes such as confusing groups with periods or misunderstanding reactivity trends. Take time to review your errors and learn from them. Consistent practice and a clear understanding of concepts will significantly improve your performance.
Practical Applications of the Periodic Table GCSE
The periodic table is not just a classroom tool—it has real-world applications in various industries. For example, elements like oxygen and hydrogen are essential for life, while metals like aluminum and iron are used in construction and manufacturing.
In medicine, elements are used in everything from diagnostic tools to treatments. For instance, radioactive isotopes are used in imaging and cancer therapy. Understanding the periodic table helps scientists develop new technologies and improve existing ones.
Even in everyday life, the periodic table plays a role. From the materials in your smartphone to the chemicals in cleaning products, elements are everywhere. This makes learning the periodic table both practical and relevant.
Conclusion
The periodic table GCSE topic is a cornerstone of chemistry education, providing a framework for understanding elements and their interactions. By learning its structure, trends, and key groups, students can develop a deeper understanding of chemical behavior.
Mastering the periodic table is not about memorization alone—it’s about recognizing patterns and applying knowledge effectively. With consistent practice and a clear understanding of concepts, students can approach their exams with confidence.
Ultimately, the periodic table is more than just a chart—it’s a powerful tool that unlocks the secrets of the chemical world. By mastering it, you set the foundation for success not only in exams but also in future scientific studies.
FAQs (Frequently Asked Questions)
What is the periodic table in GCSE Chemistry?
It is a chart that organizes elements based on atomic number and properties.
How are elements arranged in the periodic table?
They are arranged in order of increasing atomic number.
What is the difference between groups and periods?
Groups are vertical columns, while periods are horizontal rows.
Why do elements in the same group have similar properties?
Because they have the same number of outer shell electrons.
What are the most important groups to study for GCSE?
Group 1, Group 7, and Group 0 are the most important.
How can I easily remember periodic table trends?
By understanding patterns rather than memorizing facts.
What are noble gases and why are they unreactive?
They have full outer electron shells, making them stable.
How does atomic structure relate to the periodic table?
It determines the position and behavior of elements.
What are common exam questions about the periodic table?
Questions on trends, groups, and reactivity.
What is the best way to revise the periodic table for GCSE exams?
Use visual aids, practice questions, and focus on understanding trends.
You May Also Read: Indeed Jobs



